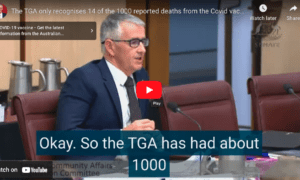
The gall of India to request actual efficacy data. That would never happen in the US.
Reuters reports that Pfizer Inc withdrew its application for emergency authorisation of its COVID-19 vaccine in India, after refusing to meet the drug regulator’s demand for additional studies.